Reconstruction of a temporal scalp defect without ipsilateral donor vessel possibilities using a local transposition flap and a latissimus dorsi free flap anastomosed to the contralateral side: a case report
Article information
Abstract
Scalp defects necessitate diverse approaches for successful reconstruction, taking into account factors such as defect size, surrounding tissue, and recipient vessel quality. This case report presents a challenging scenario involving a temporal scalp defect where ipsilateral recipient vessels were unavailable. The defect was effectively reconstructed utilizing a transposition flap and a latissimus dorsi free flap, which was anastomosed to the contralateral recipient vessels. Our report underscores the successful reconstruction of a scalp defect in the absence of ipsilateral recipient vessels, emphasizing the importance of employing appropriate surgical interventions without necessitating vessel grafts.
INTRODUCTION
Scalp defects may occur secondary to trauma, infections, constant pressure, or operations on nearby structures. They pose unique challenges due to the convex shape and considerations for intracranial structures and cosmetic outcomes [1]. There are many options for scalp defect reconstruction. Primary closure can be used for small-sized defects, but different approaches must be considered for larger-size defects, accounting for many factors, including local or regional flaps, skin grafting, or free flaps [2]. For extensive scalp defect reconstructions with osteonecrosis without surrounding viable tissues or implant bedding, free flaps may be the only viable option [3]. Furthermore, factors such as chronic infection, postoperative infection, large defect size and lack of suitable recipient vessels may present a challenge during reconstruction [4,5]. Vessel grafts can be used to bypass insufficient vessels and approach adequate recipient vessels [6]. However, additional surgical procedures can lead to higher failure rates. In this case report, we describe the successful reconstruction of a large temporal area defect without adjacent recipient vessels using a transposition flap, latissimus dorsi free flap, and contralateral recipient vessels.
CASE REPORT
A 42-year-old woman was referred with a skull defect in the right temporal area and soft tissue necrosis that had occurred after decompressive craniectomy (Fig. 1A). Computed tomography angiography showed total occlusion of the right external carotid artery; therefore, the ipsilateral facial artery and superficial temporal artery had no circulation (Fig. 1B). The reconstruction date was scheduled considering two factors: (1) according to the neurosurgery department, a titanium skull plate had to be implanted within three months after craniectomy; (2) necrosis of the defect site was still under progress and demarcation was needed to assess the final defect size. Although the demarcation was incomplete, the operation schedule was set at 83 days after the initial identification of the temporal defect and 113 days after craniectomy to prevent possible neurological complications. No notable abnormalities were observed at the transposition flap donor site, except for the presence of a hemicoronal incision scar line resulting from the previous decompressive craniectomy (Fig. 2A).

A 42-year-old woman with a right temporal region scalp defect. (A) An 18×6 cm defect is observed in the right temporal area 31 days after decompressive craniectomy. (B) Computed tomography shows total occlusion of the right external carotid artery (red arrow).
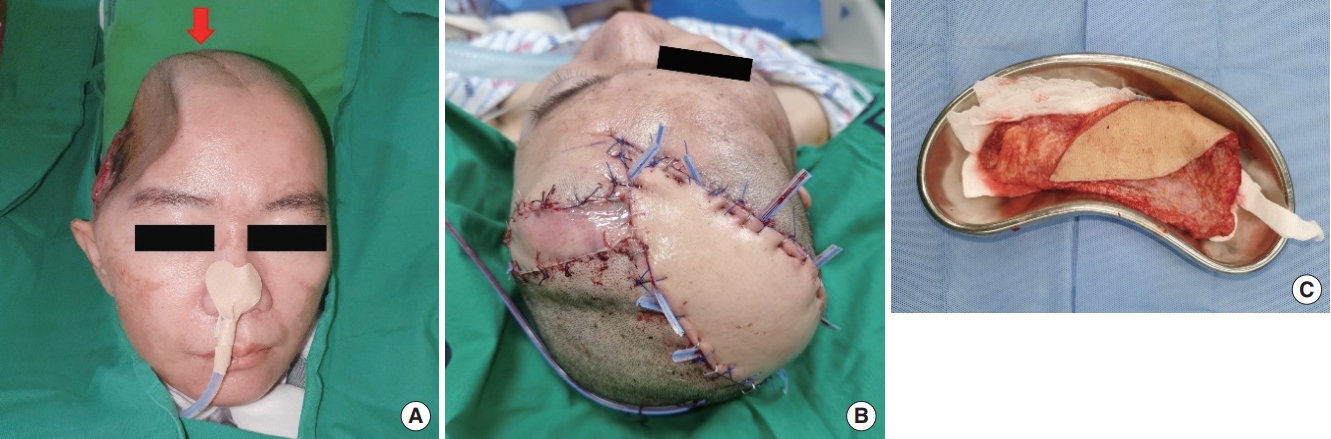
Perioperative photographs. (A) The transposition flap donor site shows a visible scar of the hemicoronal incision (red arrow). (B) An additional sagittal, linear incision is made on the scalp 12 cm away from the left superficial temporal artery for flap elevation, considering the maximal length of the latissimus dorsi free flap pedicle. The flap is transposed to the temporal area to cover the original defect, resulting in a new 18×7 cm sized defect in the fronto-parietal area. (C) The new defect is covered with a latissimus dorsi free flap using the left superficial temporal vessels as the recipient.
Prior to reconstruction, the neurosurgery team performed cranioplasty with a 3-dimensional printed titanium skull plate. The final defect size after debridement was 18× 6 cm. Extending the incision line made by the neurosurgeons, an additional sagittal, linear incision was made on the scalp 12 cm away from the contralateral superficial temporal artery for flap elevation, considering the possible maximal length of the latissimus dorsi free flap pedicle length. Next, the flap was transposed to the temporal area to cover the original defect, and the frontoparietal area was left with a new 18× 7 cm sized defect. The new defect was covered with a latissimus dorsi free flap, using the left superficial temporal vessels as the recipient. The thoracodorsal vein and artery were anastomosed to the left superficial temporal vessels in an end-to-end fashion. A 200-cc Jackson-Pratt drain was placed along the latissimus dorsi flap, and silastic drains were inserted around the flap and anastomosis site (Fig. 2B and C). Next, to prevent compression of the pedicle, a 12 × 3 cm split-thickness skin graft was harvested from the left flank and placed over the pedicle. Finally, primary closure of the donor site was performed (Fig. 3).

Postoperative photographs. (A) In order to prevent compression of the pedicle, a 12×3 cm split-thickness skin graft from the left flank is placed over the pedicle (red arrow). (B) The donor site is closed primarily.
Both the transposition flap and latissimus dorsi free flap remained stable after the reconstruction procedure. Distal flap margin revision was performed on the 16th postoperative day. The skin staplers were removed on the 19th postoperative day, and the patient was discharged on the 57th postoperative day (Fig. 4).
DISCUSSION
There are several ways to reconstruct scalp defects, and the defect size is the main deciding factor in choosing a reconstruction method. Primary closure is the standard approach for small scalp defects, whereas local flaps are the preferred option for small- to moderate-sized defects [7]. Also, a skin graft can be a viable option if a sufficient grafting bed like periosteum is available [8]. However, the patient in the case received 3-dimensional printed titanium skull plate during the cranioplasty and no periosteum was available for skin graft. Furthermore, since the scalp tissue is inelastic and lacks suitable regional flap donor sites, free flap may be the only option for the reconstruction of large, extensive scalp defects [3].
Regarding the size of the defect, the anterolateral thigh flap, deep inferior epigastric perforator flaps, latissimus dorsi flap, or other free flaps can be candidates for reconstruction of large scalp defects. The length of the anterolateral thigh flap pedicle is relatively short, ranging from 8 to 14 cm. Furthermore, when used for coverage of large defects, partial necrosis may occur [9]. Next, the deep inferior epigastric perforator flap was ruled out because of its thick and bulky nature. The latissimus dorsi flap was the preferred treatment approach in our case. According to the literature, latissimus dorsi flap pedicle can be harvested up to 18 cm, long enough to reach the contralateral recipient vessels in our case [10]. It can also provide ample tissue for extensive defect coverage. In addition, a thin, wide, and robust latissimus dorsi muscle could be harvested with the skin to provide a robust barrier against titanium foreign body bedding.
Another factor to consider is the distance between donor and recipient vessels. In cases where the pedicle length is not long enough to reach suitable recipient vessels, a vessel graft can be implemented as a direct solution. However, using a vessel graft to connect donor and recipient vessels means that additional anastomosis is inevitable. An increased number of anastomoses put the flap at a greater risk of thrombosis formation, thus leading to catastrophic failure [11]. In our case, instead of using a vessel graft to simply provide a way to reach the contralateral recipient vessels, we performed a transposition flap to resurface the original defect and created a new defect nearer to the contralateral vessels, enough for the latissimus dorsi free flap pedicle to reach recipient vessels directly.
Naturally, we also had to consider the viability of the transposition flap. Blood supply to the skin and deep tissue of the scalp is provided by the occipital, superficial temporal, and ophthalmic arteries and their perforators, respectively [12]. In this case, although the superficial temporal and ophthalmic arteries were cut off from the transposition flap, necrosis of the flap did not occur. Designing the transposition flap using the existing hemicoronal incision line alone was not enough to reduce the tension after transposition. Therefore, we had to elongate and make additional 1.5 cm incision in forehead direction. Interestingly, this case might prove that blood flow from the occipital artery alone is sufficient to supply both parietal and frontal regions of the scalp and it is safe to make additional incision pass the hairline.
In conclusion, an extensive scalp defect without adjacent ipsilateral recipient vessels due to large vessel occlusion can be successfully reconstructed using an adjacent scalp transposition flap and a latissimus dorsi free flap without an additional vessel graft.
Notes
Conflict of interest
No potential conflict of interest relevant to this article was reported.
Funding
None.
Ethical approval
The report was approved for exemption by the Institutional Review Board of Hanyang University Hospital (IRB No. HYUH 2023-02-039).
Patient consent
The patient provided written informed consent for the publication and use of her images.
Author contributions
Conceptualization: Jung Kwon An, Youn Hwan Kim, Kyunghyun Min. Data curation: Jung Kwon An, Kyunghyun Min. Formal analysis: Kyunghyun Min. Methodology: Jung Kwon An, Kyunghyun Min. Project administration: Kyunghyun Min. Writing - original draft: Jung Kwon An. Writing - review & editing: Kyunghyun Min. Investigation: Jung Kwon An, Lan Sook, Kyunghyun Min. Supervision: Seong Oh Park, Lan Sook Chang, Youn Hwan Kim, Kyunghyun Min. Validation: Seong Oh Park, Youn Hwan Kim, Kyunghyun Min.

